8 The Brain’s Reward System
Reading Objectives
By the end of this chapter, you should be able to:
- Identify the key components of the mesolimbic dopamine pathway and describe their roles in mediating reward and motivation.
- Explain the concepts of reward prediction error, “wanting” versus “liking,” and incentive sensitization as they relate to the dopamine system and addictive behaviors.
- Analyze how the opponent-process theory illustrates the balance between pleasure and pain, and how repeated substance use shifts the hedonic set-point toward dysphoria and craving.
- Evaluate the impact of chronic drug use on neural circuitry, with attention to neuroadaptations such as receptor downregulation and impaired prefrontal control.
- Assess how neuroimaging techniques such as fMRI and PET reveal functional and neurochemical changes in the mesolimbic dopamine pathway associated with addiction.
Key Terms
- Mesolimbic Dopamine Pathway: A neural circuit that originates in the ventral tegmental area (VTA) and projects to limbic regions such as the nucleus accumbens. It plays a central role in reward processing and motivation. Here, “reward” is a latent construct. It is an internal motivational state inferred from behavior and neural signals rather than directly observed.
- Dopamine: A neurotransmitter involved in reinforcement learning and motivation. Dopamine signals the importance of outcomes and cues, shaping future behavior by influencing pursuit and learning rather than pleasure alone.
- Ventral Tegmental Area (VTA): A midbrain region that produces dopamine and serves as a primary source of dopaminergic input to the mesolimbic pathway.
- Nucleus Accumbens (NAc): A structure in the ventral striatum that receives dopamine from the VTA and integrates information about reward, motivation, and goal-directed action.
- Reward Prediction Error (RPE): The difference between what is expected and what actually occurs. Dopamine increases or decreases signal whether an outcome is better or worse than anticipated, updating learning.
- Incentive Sensitization: A process in which repeated drug exposure heightens the motivational pull or “wanting” of substance-related cues, even when the pleasurable effects or “liking” diminish.
- Opponent-Process Theory: A model describing how initial pleasurable responses to a stimulus are followed by an opposing process that produces discomfort or stress, shifting the balance between reward and pain over time.
- “Wanting” vs. “Liking”: The distinction between dopamine-driven motivation to seek a reward (“wanting”) and the hedonic pleasure experienced during consumption (“liking”).
- Functional Magnetic Resonance Imaging (fMRI): An imaging technique that measures brain activity through changes in blood flow, allowing researchers to study functional neural circuits during tasks or at rest.
- Positron Emission Tomography (PET): An imaging method that uses radioactive tracers to visualize and quantify neurochemical processes such as dopamine receptor availability and neurotransmitter release.
Introduction
The brain constantly balances pleasure and pain, much like a seesaw. Dopamine is often described as the brain’s “molecule of more” because it drives motivation and pursuit rather than satisfaction itself. When something rewarding occurs, dopamine helps reinforce the behaviors and cues that led to it.
After pleasure, however, the brain activates an opposing response that produces stress or dysphoria. This dynamic is described by opponent-process theory. Early on, the pleasurable effects outweigh the discomfort. With repeated pursuit of rewards without sufficient balance, whether from substances, screens, or highly palatable foods, the opposing process grows stronger. Over time, the “pain side” begins to dominate.
As this shift unfolds, people may find that rewards no longer produce the same enjoyment. Instead, they are pursued to relieve discomfort or to feel normal. This gradual change in the hedonic set-point, moving from pleasure toward dysphoria and craving, is a defining feature of addiction and a central focus of this chapter.
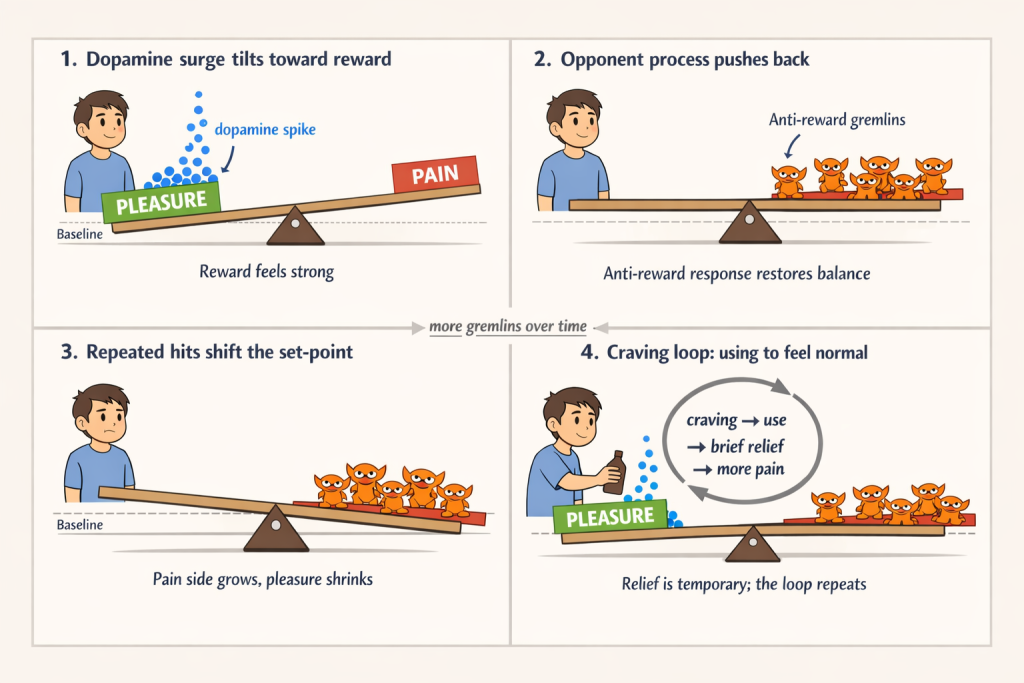
In the brain’s intricate machinery, pleasure and pain sit on opposite ends of a delicate seesaw, a metaphor popularized by Anna Lembke (Lembke, 2021). When we experience pleasure, dopamine surges tip the balance toward reward. That surge then triggers a compensatory “anti-reward” response, often illustrated as small stress-inducing forces piling onto the pain side of the seesaw. With repeated exposure, these anti-reward forces accumulate, shifting the balance toward pain and driving persistent craving.
In this module, we explore how the limbic–basal ganglia pathway underlies this transformation. You will learn the crucial distinction between “wanting,” the dopamine-driven urge to seek rewards, and “liking,” the actual pleasure derived from them. We examine how chronic substance use reshapes neural circuits, locking in compulsive habits and intense cravings long after the initial euphoria fades. We also show how modern brain-imaging methods reveal these deep-seated changes, reinforcing the view that addiction reflects fundamental neurobiological processes rather than a simple failure of willpower.
A Quick Overview of the Human Brain
Before describing the reward pathway, it helps to establish some basic vocabulary. The brain coordinates chemical and electrical signals that regulate everything from life-sustaining functions like breathing and digestion to perception, emotion, thought, and social interaction. The human brain contains roughly 86 billion nerve cells, called neurons, along with a variety of supporting cells (SAMHSA, 2016).
Each neuron has three main components. The cell body houses the nucleus and directs the neuron’s activities. The axon is a long projection that sends signals to other cells. The dendrites are branching structures that receive incoming signals from neighboring neurons. Together, these components form vast networks that allow information to flow through the brain, setting the stage for understanding how reward, motivation, and addiction emerge from neural circuitry.
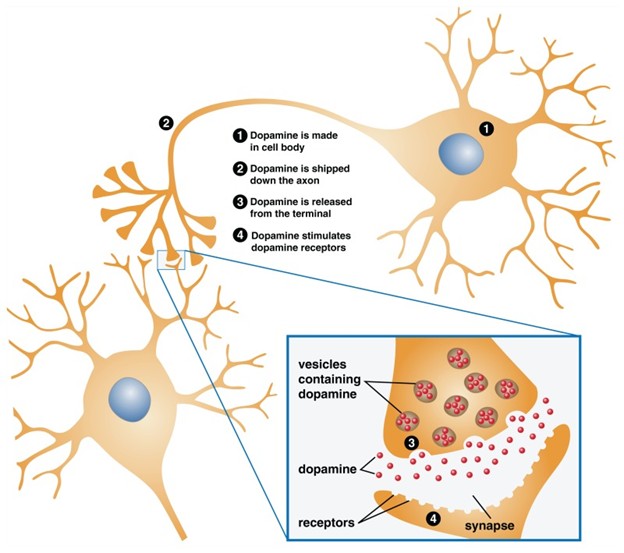
Neurons communicate using chemical messengers called neurotransmitters. These chemicals cross a tiny gap between cells, known as a synapse, and bind to receptors on neighboring neurons. Some neurotransmitters inhibit or dampen activity in the receiving neuron, making it less likely to fire. Others are excitatory, increasing the likelihood that the neuron will pass the signal along. Behavior and cognition emerge from the balance of these excitatory and inhibitory influences across large networks of neurons.
Neurons are not wired randomly. They tend to cluster into specialized circuits that carry out particular functions. Some circuits support higher-order processes such as thinking, learning, emotion, and memory. Others are more directly tied to action, linking the brain to muscles to produce movement, or to sensory systems that process information from the eyes, ears, and skin. Addiction-related processes arise not from a single “addiction center,” but from interactions among multiple circuits that normally support motivation, learning, and self-control.
Core Neuroanatomy of the Mesolimbic Dopamine Pathway
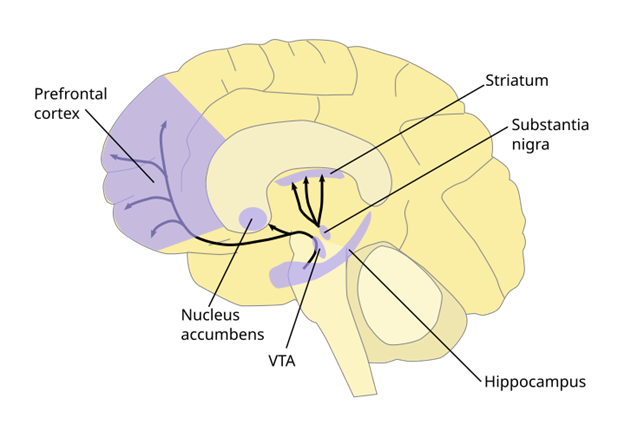
The mesolimbic dopamine pathway has two central hubs. The ventral tegmental area (VTA) is a cluster of dopamine-producing neurons located in the midbrain. The nucleus accumbens (NAc) sits in the ventral striatum and acts as a key integration site for reward, motivation, and learning signals.
When outcomes are better than expected, dopamine neurons in the VTA fire in brief bursts, releasing dopamine into the nucleus accumbens. When outcomes are worse than expected, these neurons reduce or pause their firing. This rise or fall in dopamine is not just a pleasure signal. It functions as a teaching signal, updating the brain about whether predictions were accurate and guiding future behavior. In this sense, dopamine is the core “currency” of learning in the mesolimbic pathway.
Although the VTA and NAc are the central actors, they do not work in isolation. Regions such as the prefrontal cortex, amygdala, hippocampus, and extended amygdala interact with this pathway to shape decision-making, emotional responses, memory for reward-related cues, and the formation of habits. These interactions become especially important for understanding cue-driven craving and compulsive behavior, which we will unpack in more detail in Module 9.
Function of the Mesolimbic Dopamine Pathway
Reward Prediction Error
Recall that dopamine neurons in the ventral tegmental area (VTA) send signals to the nucleus accumbens (NAc) and other limbic and cortical structures. When rewards exceed expectations, these VTA neurons increase dopamine release, reinforcing the behaviors that led to the positive outcome. This raises a deeper question. Why does dopamine fire, and what information is it conveying?
Dopamine neurons signal a reward prediction error (RPE), which reflects how actual outcomes compare with expectations. If a reward is better than expected, such as studying for a B and receiving an A+, dopamine neurons show a brief surge in activity. That surge reinforces the actions that led to the unexpectedly good result (Schultz, 1998). In contrast, when outcomes are worse than expected, dopamine neuron firing is reduced, guiding the brain to revise future behavior.
This RPE mechanism functions as a teaching signal. A dopamine spike communicates, “This was better than expected. Strengthen whatever led here.” A dopamine dip communicates, “This outcome fell short. Adjust your strategy.” Across repeated experiences, these signals help the brain learn which cues, contexts, and actions reliably lead to rewarding outcomes.
Importantly, reward prediction errors shape not only learning but also motivation. As cues become associated with dopamine surges, they can acquire powerful motivational pull. This helps explain why people may pursue rewards intensely, even when the actual experience delivers less pleasure than anticipated.

This reward-prediction mechanism is the brain’s natural version of what computer scientists call reinforcement learning, a process by which agents learn to maximize future rewards through feedback. Dopamine’s prediction-error signals continuously update the brain’s internal “value map,” strengthening behaviors that produce better-than-expected outcomes and weakening those that disappoint. This learning logic brings us to a critical distinction in the dopamine system: the difference between wanting and liking.
Wanting vs. Liking
Dopamine primarily drives “wanting”—the motivational force that energizes pursuit of rewards—more than “liking,” the pleasure experienced when a reward is consumed (Berridge & Robinson, 2016). A useful mechanistic label for cue-triggered wanting is incentive salience: the process by which reward-related cues become motivational magnets that grab attention and energize approach behavior. These processes are dissociable in the brain. Dopamine-related signaling is strongly tied to incentive motivation/craving and approach behavior, while “liking” depends more heavily on opioid-based hedonic hotspots that generate pleasure.
Both wanting and liking are latent constructs: we do not observe them directly. Instead, we infer them from patterns of behavior (e.g., approach, effort, reaction time), self-report (e.g., craving ratings), and neural measures that serve as proxies for circuit engagement. In cue-driven situations, increased wanting is often inferred when cues draw attention and trigger approach/effort—consistent with increased incentive salience—even if the pleasurable experience of consumption does not increase.
A simple illustration makes the distinction concrete. Imagine you have a strong preference for chocolate cake. Seeing the cake in a display case can trigger intense wanting: your attention locks in, your mouth waters, and you feel a pull toward eating it. In incentive-salience terms, the cue (the sight of cake) has been tagged with motivational power. Now imagine taking a bite. The rich, chocolate flavor produces enjoyment. That pleasure is liking. Importantly, wanting can grow stronger even as liking stays the same or declines—a divergence that becomes central to understanding addiction.
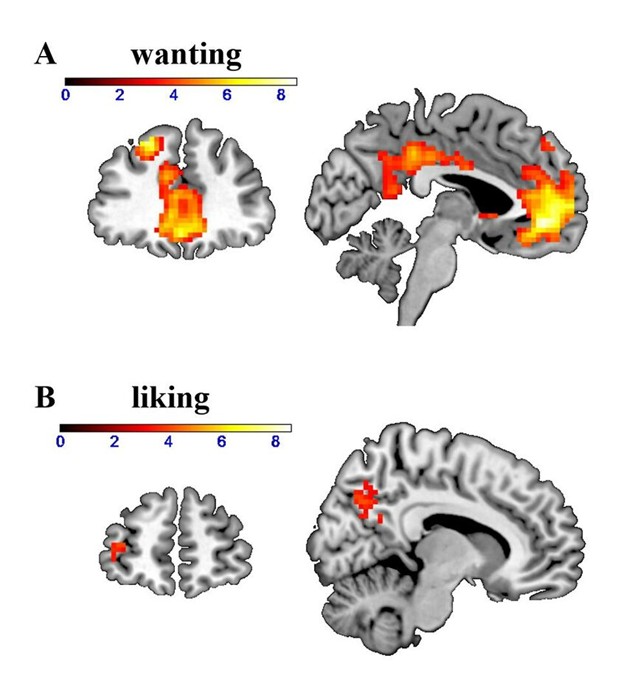
This wanting–liking split is formalized in the incentive-sensitization theory of addiction.
Incentive-Salience Hypothesis
The incentive-salience hypothesis proposes that the brain can tag reward-related cues (or mental representations of rewards) with motivational power, so those cues come to trigger strong wanting. In this view, cues become motivational magnets that grab attention and energize approach behavior even when the actual pleasure of consumption (liking) is unchanged or reduced (Berridge & Robinson, 2016). Incentive sensitization refers to the long-term increase in the brain’s propensity to assign incentive salience to drug cues after repeated drug use—helping explain intense cue-evoked craving and relapse risk even when the drug is no longer very pleasurable.
In substance use disorders, dopamine-linked wanting can grow increasingly intense even as liking declines. This pattern reflects incentive sensitization (cues trigger exaggerated incentive salience) alongside tolerance (reduced pleasure from the substance itself). The result is powerful craving without proportional enjoyment.
Neuroanatomy helps explain this divergence. Dopamine-related signaling in the nucleus accumbens (NAc), especially the core, is strongly implicated in cue-triggered wanting and approach behavior. In contrast, hedonic liking is mediated in part by opioid-based “hedonic hotspots,” including regions in and around the NAc shell. Because these systems are partially separable, craving can intensify even as pleasure fades—one of the clearest signatures of addiction.

The Pleasure–Pain Balance
While reward prediction errors fine-tune behavior by comparing outcomes to expectations, the brain also works to maintain emotional stability through homeostatic regulation. Homeostasis refers to the self-regulating processes that keep biological systems within functional ranges while adapting to changing conditions. After pleasurable experiences, dopamine surges are typically followed by brief dips in mood or motivation. This counter-response restores equilibrium, much like a seesaw tipping back after a high.
This pleasure–pain balance keeps motivation calibrated. It allows us to enjoy rewards and learn from them without becoming trapped in constant highs or immobilized by lows. Under typical conditions, the system resets efficiently, preserving sensitivity to everyday rewards.
Problems arise when dopamine spikes are repeated and intense, as with addictive drugs or highly stimulating behaviors. Over time, the brain adapts by reducing dopamine sensitivity and increasing anti-reward or stress-related signals. Natural rewards begin to feel less satisfying, while baseline stress and irritability increase. The individual is no longer chasing pleasure alone but is increasingly motivated to escape discomfort.
In this way, a system designed to support learning and balance can be pushed into dysregulation. Repeated overstimulation drives cycles of craving, tolerance, and dysphoria. Although the specific neuroadaptations vary by substance, they share a common core: a shifted hedonic balance in which the brain’s reward system is recalibrated toward pain rather than pleasure.
Substance-Specific Effects on the Mesolimbic System
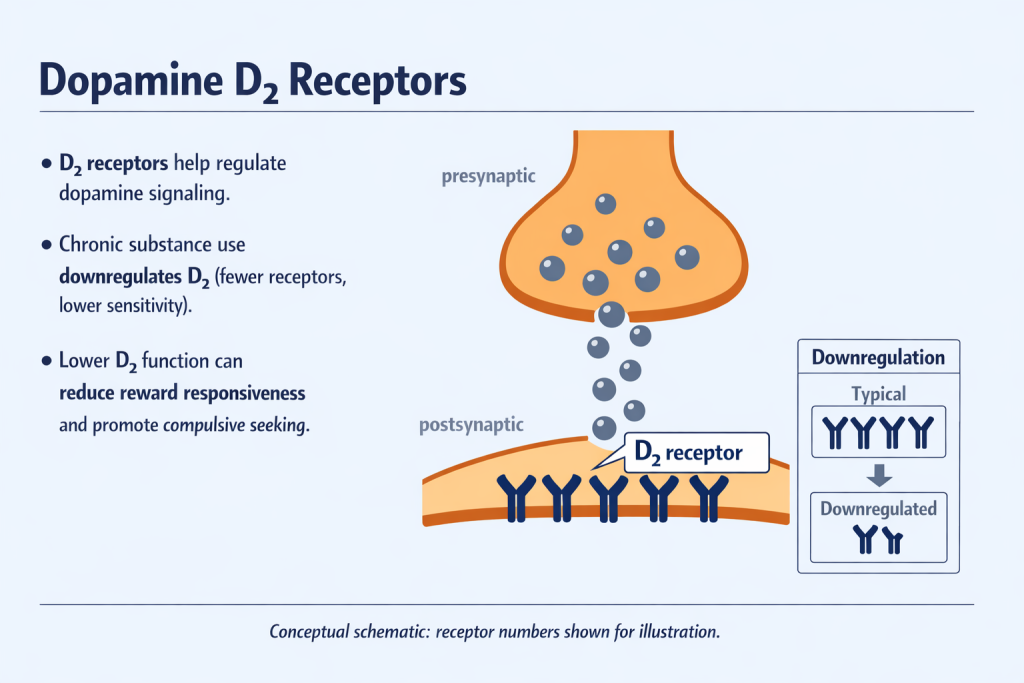
Initially, drugs trigger exaggerated dopamine spikes, producing large positive reward prediction errors. These surges create intense feelings of pleasure and strongly reinforce substance use. With repeated exposure, however, the brain adapts. Neuroadaptations such as receptor downregulation, reduced dopamine sensitivity, and increased anti-reward signaling, including systems involving dynorphin and corticotropin-releasing factor (CRF), begin to dominate. The result is a shift in the brain’s pleasure–pain balance toward pain.
As this balance shifts, motivation changes. Behavior is no longer driven primarily by the pursuit of pleasure but by the need to avoid withdrawal, stress, and dysphoria (Lembke, 2021). What once felt rewarding becomes necessary simply to feel normal.
In essence, addiction reflects a corrupted version of the brain’s natural reward system. Pathological craving, or excessive “wanting,” persists even as genuine enjoyment, or “liking,” diminishes. Although this core process is shared across substances, the precise neurobiological mechanisms and downstream consequences vary depending on the drug involved.

| Substance | Mechanism of Dopamine Activation | Dopamine Impact | Withdrawal Profile | Key Neuroadaptations |
|---|---|---|---|---|
| Alcohol | Disinhibits VTA dopamine neurons via GABA modulation; increases endogenous opioids | Moderate dopamine increase; enhanced initial euphoria | Moderate to severe: anxiety, anhedonia, irritability | ↓ D₂ receptors, ↑ CRF (stress), impaired frontal cortex function |
| Cannabis | THC activates CB₁ receptors → disinhibits dopamine in VTA | Mild to moderate dopamine increase | Mild: irritability, sleep disruption, low motivation | ↓ dopamine release (long-term), blunted response to rewards |
| Opioids | Inhibit GABA interneurons → disinhibit VTA dopamine neurons; direct activation of opioid receptors | Strong dopamine surge plus direct hedonic effects | Severe: dysphoria, physical pain, intense craving | ↓ D₂ receptors, ↓ endogenous opioid tone, cue-induced dopamine sensitization |
| Cocaine | Blocks dopamine reuptake → large synaptic dopamine accumulation | Very strong, rapid dopamine spike | High craving, mood crashes, anhedonia | ↓ D₂ receptors, ↓ dopamine release capacity, strong cue reactivity |
Below is an overview of how common substances—alcohol, cannabis, opioids, and cocaine—affect the mesolimbic dopamine system. Although their pharmacological mechanisms differ, all ultimately distort dopamine signaling in ways that promote craving, tolerance, and dependence.
Alcohol
Alcohol initially boosts dopamine release by disinhibiting dopamine neurons in the ventral tegmental area (VTA). This occurs primarily through suppression of inhibitory GABA neurons and increased release of endogenous opioids, producing mild euphoria and relaxation (Ludlow et al., 2009).
With chronic use, the brain adapts by reducing dopamine receptor availability, particularly D₂ receptors, which blunts pleasure from both alcohol and natural rewards. During withdrawal, stress systems become overactive, producing anxiety, dysphoria, and irritability. These aversive states strongly motivate continued drinking as a form of negative reinforcement. Long-term alcohol misuse also impairs prefrontal cortex functioning, contributing to impulsivity, poor decision-making, and heightened craving (Hienz et al., 2004).
Cannabis
Δ9-tetrahydrocannabinol (THC), the primary psychoactive compound in cannabis, increases dopamine release indirectly by activating CB₁ receptors on GABA interneurons in the VTA, thereby disinhibiting dopamine neurons (Bloomfield et al., 2016). The resulting dopamine increase is typically smaller than that produced by stimulants or opioids.
With heavy or chronic use, cannabis is associated with reduced dopamine responsiveness, leading to a hypodopaminergic state that may underlie reduced motivation and reward sensitivity, sometimes described as an amotivational profile. PET imaging studies show diminished dopamine release capacity in frequent cannabis users, even though physical withdrawal symptoms are generally mild (Bloomfield et al., 2016). Notably, the cannabinoid system remains a promising therapeutic target, particularly through non-intoxicating compounds such as cannabidiol (CBD).
Opioids (Heroin, Morphine, Prescription Painkillers)
Opioids are highly addictive because they act on the reward system through two converging mechanisms. First, they directly activate μ-opioid receptors, producing strong hedonic effects. Second, they indirectly increase dopamine release by inhibiting GABA interneurons in the VTA, disinhibiting dopamine neurons (Hou et al., 2012).
Chronic opioid exposure leads to pronounced reductions in dopamine receptor availability and overall dopamine release capacity. At the same time, the dopamine system becomes sensitized to drug-related cues, producing intense craving that can persist long after abstinence. Withdrawal is marked by severe dysphoria, physical pain, and stress, illustrating the extreme pleasure-to-pain shift characteristic of opioid addiction. Pharmacological treatments such as methadone and buprenorphine stabilize opioid signaling and reduce withdrawal and craving, supporting recovery by dampening reward-system volatility (Hou et al., 2012).
Cocaine and Other Stimulants
Stimulants such as cocaine produce rapid and intense dopamine spikes by blocking dopamine reuptake, causing dopamine to accumulate in synapses (Volkow & Morales, 2015). The magnitude and speed of this dopamine elevation are uniquely high, making stimulants especially reinforcing.
Repeated stimulant use triggers rapid dopamine receptor downregulation, particularly of D₂ receptors, and a marked decline in baseline dopamine responsiveness. Chronic cocaine users often experience profound anhedonia, low motivation, and emotional flattening when not using the drug. Cue-induced craving becomes dominant, overpowering natural reward-seeking behaviors. Recovery of dopamine function can take months or years, contributing to high relapse risk. Behavioral treatments, including contingency management, remain central to care and aim to support gradual restoration of reward-system balance (Volkow & Morales, 2015).
While each substance affects the mesolimbic dopamine pathway in distinct ways, they converge on a common outcome: distorted dopamine signaling that transforms healthy reward seeking into pathological craving and dependence. Across drugs, exaggerated dopamine responses, compensatory neuroadaptations, and heightened anti-reward signaling reshape motivation so that behavior becomes organized around relief from discomfort rather than the pursuit of pleasure.
Understanding these mechanisms has practical and ethical importance. It informs more effective prevention and treatment strategies, and it supports a compassionate, evidence-based view of addiction as a neurobiological disorder rather than a moral or personal failing.
Having examined how substances alter dopamine pathways, the next logical step is to explore how scientists visualize these changes in the living human brain. This brings us to the field of neuroimaging.
Digital Dopamine: Social Media and the Mesolimbic Pathway
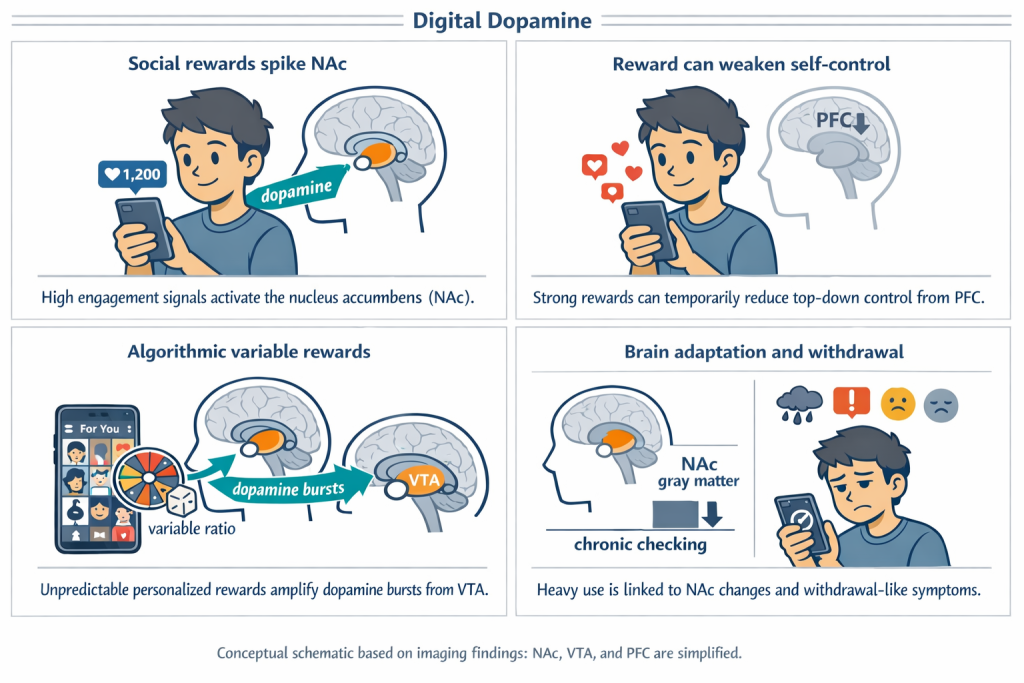
Variable social rewards keep us scrolling. Social media interactions, such as Instagram “Likes,” follows, and TikTok views, function as potent yet unpredictable social rewards. Brain imaging studies show that when adolescents view images with many “Likes,” there is increased activation in the nucleus accumbens (NAc), the same dopamine-rich region engaged by food, drugs, and money (Sherman et al., 2016). Stronger accumbens responses to social reward predict more frequent checking of social media feeds, reinforcing a habit loop that parallels substance addiction (Sherman et al., 2018).
Algorithms amplify dopamine-driven reward cycles. Platforms like TikTok intensify this process through algorithmic curation. When participants view personalized content on TikTok’s “For You” page, fMRI studies show greater activation of the ventral tegmental area (VTA), the midbrain source of dopamine signaling, compared to generic feeds (Lin et al., 2021). This variable-ratio schedule of unpredictable but personalized rewards promotes persistent engagement and compulsive scrolling.
Chasing the digital hit reshapes the brain. Chronic social media engagement does not merely activate reward circuits in the moment. Smartphone usage studies show that frequent Instagram and Facebook checking is associated with reduced gray matter volume in the nucleus accumbens, a pattern similar to structural changes observed in long-term substance use (Montag et al., 2017).
The pleasure–pain seesaw applies without chemicals. Heavy social media users often report withdrawal-like symptoms, including irritability, anxiety, and low mood, during periods of digital abstinence. These effects mirror the opponent-process imbalance described earlier in this module, demonstrating that powerful dopamine-driven reward loops can emerge even in the absence of psychoactive substances (Lembke, 2021).
Imaging the Dopamine Pathway: fMRI, PET, and the MID Task
How do we know so much about the dopamine system in humans without directly measuring neuron activity? Two advanced neuroimaging techniques—functional Magnetic Resonance Imaging (fMRI) and Positron Emission Tomography (PET)—allow scientists to observe how dopamine pathways operate in real time.
Functional Magnetic Resonance Imaging (fMRI) is a specialized type of MRI that detects brain activity indirectly by measuring changes in blood flow. MRI scanners are large, cylindrical devices that use powerful magnets and radio waves to create detailed images of the brain. During an fMRI scan, participants lie on a sliding table that moves into the scanner’s tunnel-like center, where changes in blood oxygenation are captured and reconstructed into three-dimensional images of brain activity.
Figure 10: The ABCD study MRI training video for ABCD participants.
Resting-State fMRI: Mapping the Brain’s Default Connectivity
What is resting-state fMRI?
Resting-state fMRI measures brain activity while a person lies quietly in the scanner without performing a specific task. Rather than capturing responses to stimuli, it records spontaneous, synchronized fluctuations in the blood-oxygen-level–dependent (BOLD) signal across brain regions. These coordinated fluctuations reflect intrinsic functional connectivity, meaning how strongly different regions communicate with one another at rest. The ABCD Study includes resting-state fMRI data for participants across development.
Why is this useful for addiction research?
Resting-state fMRI is particularly valuable for identifying baseline differences in brain network organization between individuals with substance use disorders and healthy controls. Across studies, addiction is associated with altered connectivity within reward and control circuits.
Key finding: reward–control disconnection.
A consistent result is disrupted functional connectivity between the nucleus accumbens (NAc), a core reward-processing region, and the prefrontal cortex (PFC), which supports executive control and decision-making. For example, stimulant users often show reduced NAc–PFC connectivity, indicating weakened top-down regulation of reward-driven impulses by frontal control systems (Sutherland et al., 2012).
Interpretation.
These patterns suggest that addiction involves not only heightened reward sensitivity, but also impaired communication between motivational systems and self-regulatory control networks. Resting-state findings therefore provide neural evidence for compulsive drug-seeking as a circuit-level disorder, not simply a failure of willpower.
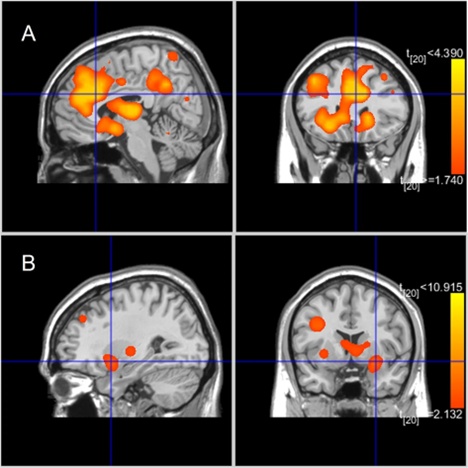
Figure 11 is a real resting-state fRMI data visualization; it is not an image of a single person’s brain, but a summary based on brain scans from multiple studies and participants (Tolomeo & Yu 2022). It visualizes group differences in resting-state brain activity between individuals with substance use and behavioral addictions (SUD + BA) and healthy controls (HC).
• Panel A (top row): Brain regions with increased connectivity in the SUD + BA group compared to controls. These include areas like the amygdala, thalamus, midbrain, caudate, and parahippocampal gyrus—all part of circuits involved in emotion, motivation, and habit learning.
• Panel B (bottom row): Brain regions with decreased connectivity in the SUD + BA group, such as parts of the posterior lobe and parahippocampal gyrus, which are linked to memory and sensory processing.
The color scale (yellow to red) reflects the strength of these group differences in connectivity. These patterns suggest that addiction is associated with hyperconnectivity in reward and habit-related brain systems and disrupted connectivity in regions important for memory and self-regulation.
Task-Based fMRI – The Monetary Incentive Delay (MID) Task
Task-based fMRI involves participants performing specific activities designed to engage particular neural circuits. A widely used paradigm in addiction research is the Monetary Incentive Delay (MID) task. Participants see cues indicating potential monetary gains or losses, prompting anticipation and engagement of reward systems.
The MID task differentiates between anticipation (expecting a reward) and feedback (receiving the reward). Crucially, dopamine neurons primarily fire during reward anticipation, reflecting “wanting,” rather than during reward receipt, reflecting “liking.” Consistent with this, fMRI studies using the MID task reveal increased activation in the NAc during anticipation phases (Knutson et al., 2000). In addiction, MID task findings commonly demonstrate abnormal activation patterns: addicted individuals typically exhibit heightened NAc responses to drug-related cues and diminished activation to natural rewards, indicating altered reward processing.
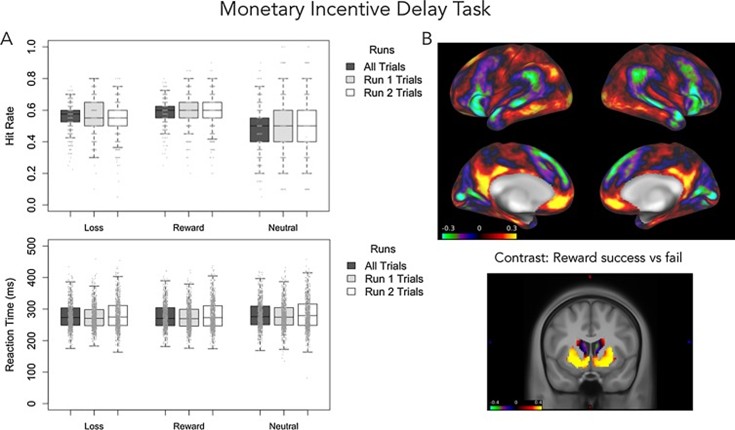
Figure 12 shows the preliminary results from the ABCD study’s baseline data collection of MID fMRI data.
Panel A (left side): Behavioral performance during the MID task
- Top graph (Hit Rate): Shows how often participants successfully responded (“hit”) to targets when they could lose money (“Loss”), win money (“Reward”), or had no monetary consequence (“Neutral”). Higher values indicate better performance.
- Bottom graph (Reaction Time): Shows how quickly (in milliseconds) participants responded to these same targets. Lower values mean faster reactions.
Panel B (right side): Brain activation during the MID task
- Top images (Surface brain maps): Illustrate overall activation patterns across the brain surface during the task, highlighting regions engaged by reward anticipation and processing.
- Bottom image (Contrast image): Specifically highlights brain areas more activated when successfully winning money (“reward success”) compared to not receiving money (“reward fail”). Warm colors (yellow/red) indicate stronger activation in the ventral striatum and nucleus accumbens, areas central to dopamine-related reward processing.
These images show that performance and brain activation during reward-related tasks like the MID are measurable using behavioral data and fMRI, allowing researchers to investigate the brain’s reward circuitry in detail.
PET Imaging: Neurochemical Insights into Dopamine
While fMRI measures brain activity indirectly through blood flow changes, PET imaging directly assesses neurochemistry by using radioactive tracers that bind to specific molecules. A widely used PET tracer, [11C]-raclopride, binds to dopamine D₂ receptors. Raclopride’s binding decreases when dopamine is released (since dopamine displaces raclopride), providing an indirect measure of dopamine release and receptor availability. PET imaging is not included in ABCD data collection.
Studies employing PET consistently find reduced D₂ receptor availability in the striatum of individuals with substance use disorders, including cocaine, alcohol, and opioid dependence (Volkow et al., 2009). Lower D₂ receptor levels are associated with increased craving, impaired self-control, and higher relapse rates, reflecting a blunted dopamine reward system.
Combining PET with fMRI provides complementary insights. For instance, individuals showing reduced NAc activation during reward anticipation in fMRI typically also demonstrate decreased D₂ receptor availability in PET, reinforcing the neurochemical underpinning of reward dysfunction in addiction.
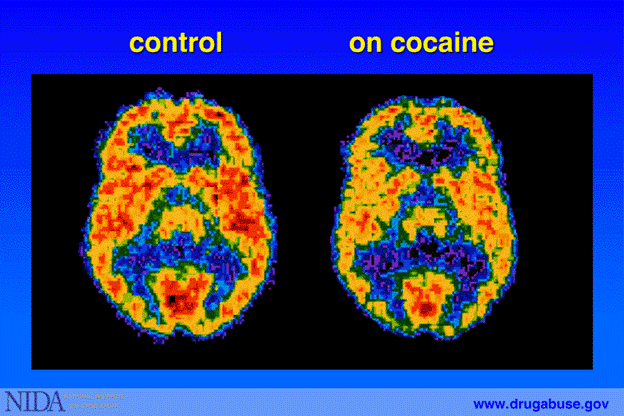
Figure 13 is a PET image that uses radiolabeled glucose to visualize metabolic activity across the brain. Red areas represent high glucose uptake, meaning active brain function, while blue and black indicate low activity. The scan on the right shows that in a person on cocaine, much of the brain exhibits reduced glucose metabolism—particularly in frontal regions—indicating that cocaine impairs normal brain function and energy use, which can contribute to disrupted thinking, poor impulse control, and long-term cognitive deficits.
Conclusion: Beyond Reward—From Motivation to Control
The mesolimbic dopamine pathway, our core reward circuit, does not function in isolation—it interacts dynamically with multiple neural networks that govern self-control, emotional regulation, habit formation, and decision-making. As we have explored in this module, dopamine fuels the “go” system that drives our pursuit of rewarding experiences. However, balanced behavior relies equally on the brain’s “stop” system—primarily involving frontal regions like the prefrontal cortex (PFC) and anterior cingulate cortex (ACC)—that evaluates consequences and modulates impulsive actions. In Module 9, we will dive deeper into these interconnected circuits.
References
Berridge, K. C., & Robinson, T. E. (2016). Liking, wanting, and the incentive-sensitization theory of addiction. American Psychologist, 71(8), 670–679. https://doi.org/10.1037/amp0000059
Bloomfield, M. A. P., Ashok, A. H., Volkow, N. D., & Howes, O. D. (2016). The effects of Δ⁹-tetrahydrocannabinol on the dopamine system. Nature, 539(7629), 369–377. https://doi.org/10.1038/nature20153
Casey, B. J., Cannonier, T., Conley, M. I., Cohen, A. O., Barch, D. M., Heitzeg, M. M., … & Dale, A. M. (2018). The adolescent brain cognitive development (ABCD) study: Imaging acquisition across 21 sites. Developmental Cognitive Neuroscience, 32, 43–54. https://doi.org/10.1016/j.dcn.2018.03.001
Heinz, A., Siessmeier, T., Wrase, J., Hermann, D., Klein, S., Grüsser, S., … & Bartenstein, P. (2004). Correlation between dopamine D(2) receptors in the ventral striatum and central processing of alcohol cues and craving. American Journal of Psychiatry, 161(10), 1783–1789. https://doi.org/10.1176/ajp.161.10.1783
Hou, H., Tian, M., & Zhang, H. (2012). Positron emission tomography molecular imaging of dopaminergic system in drug addiction. The Anatomical Record, 295(5), 722–733. https://doi.org/10.1002/ar.22430
Knutson, B., Westdorp, A., Kaiser, E., & Hommer, D. (2000). FMRI visualization of brain activity during a monetary incentive delay task. NeuroImage, 12(1), 20–27. https://doi.org/10.1006/nimg.2000.0593
Lembke, A. (2021). Dopamine Nation: Finding Balance in the Age of Indulgence. New York, NY: Dutton.
Ludlow, K., Bradley, K., Allison, D., Taylor, S., Yorgason, J., Hansen, D., … & Steffensen, S. (2009). Acute and chronic ethanol modulate dopamine D2-subtype receptor responses in ventral tegmental area GABA neurons. Alcoholism: Clinical and Experimental Research, 33(5), 804–811. https://doi.org/10.1111/j.1530-0277.2009.00899.x
Montag, C., Markowetz, A., Błaszkiewicz, K., Andone, I., Lachmann, B., Sariyska, R., Trendafilov, B., Eibes, M., Kolb, J., Reuter, M., & Weber, B. (2017). Facebook usage on smartphones and gray matter volume of the nucleus accumbens. Behavioural Brain Research, 329, 221–228. https://doi.org/10.1016/j.bbr.2017.04.035
National Institute on Drug Abuse. (2012). Dopamine pathways [Diagram]. Wikimedia Commons. https://commons.wikimedia.org/wiki/File:Dopamine_pathways.jpg
SAMHSA. (2016). Facing Addiction in America: The Surgeon General’s Report on Alcohol, Drugs, and Health. U.S. Department of Health and Human Services. https://www.ncbi.nlm.nih.gov/books/NBK424849/
Schultz, W. (1998). Predictive reward signal of dopamine neurons. Journal of Neurophysiology, 80(1), 1–27. https://doi.org/10.1152/jn.1998.80.1.1
Sherman, L. E., Greenfield, P. M., Hernandez, L. M., & Dapretto, M. (2018). Peer influence via Instagram: Effects on brain and behavior in adolescence and young adulthood. Child Development, 89(1), 37–47. https://doi.org/10.1111/cdev.12838
Sherman, L. E., Payton, A. A., Hernandez, L. M., Greenfield, P. M., & Dapretto, M. (2016). The power of the like in adolescence: Effects of peer influence on neural and behavioral responses to social media. Psychological Science, 27(7), 1027–1035. https://doi.org/10.1177/0956797616645673
Soutschek, A., Tobler, P. N., Kahnt, T., Quednow, B. B., & Weber, S. C. (2021). Opioid antagonism reduces wanting by strengthening frontostriatal connectivity. bioRxiv. https://doi.org/10.1101/2021.06.20.449203
Su, C., Zhou, H., Gong, L., Teng, B., Geng, F., & Hu, Y. (2021). Viewing personalized video clips recommended by TikTok activates default mode network and ventral tegmental area. NeuroImage, 237, 118136. https://doi.org/10.1016/j.neuroimage.2021.118136
Tolomeo, S., & Yu, R. (2022). Brain network dysfunctions in addiction: A meta-analysis of resting-state functional connectivity. Translational Psychiatry, 12(1), 41. https://doi.org/10.1038/s41398-022-01792-6
Volkow, N. D., & Morales, M. (2015). The brain on drugs: From reward to addiction. Cell, 162(4), 712–725. https://doi.org/10.1016/j.cell.2015.07.046
Volkow, N. D., Fowler, J. S., Wang, G. J., Baler, R., & Telang, F. (2009). Imaging dopamine’s role in drug abuse and addiction. Neuropharmacology, 56(Suppl 1), 3–8. https://doi.org/10.1016/j.neuropharm.2008.05.022

