9 Cravings & Control
Reading Objectives
-
Identify key brain regions involved in craving and control, including amygdala, hippocampus, nucleus accumbens, extended amygdala, prefrontal cortex, and basal ganglia, and describe their functions.
-
Explain how classical conditioning turns neutral cues and contexts into triggers for cue-induced craving, including emotional tagging and contextual memory.
-
Describe how stress and withdrawal amplify craving through the extended amygdala and negative reinforcement.
-
Describe how the basal ganglia “Go” and “No-Go” pathways and dopamine bias regulate action selection, habit formation, and impulse control.
-
Integrate limbic motivation and executive control to explain why some cue-triggered urges become actions while others are inhibited, and how chronic substance use shifts the balance toward automatic habits and weaker control.
-
Recognize how researchers measure these processes using task fMRI (cue reactivity, SST, n-back), structural MRI, DTI, and behavioral assessments of inhibition and decision-making.
Key Terms
- Amygdala: An almond-shaped limbic structure that tags experiences with emotional significance (e.g. linking a cue with a feeling).
- Hippocampus: A seahorse-shaped brain region for forming and retrieving contextual and spatial memories (the “where and when” of events).
- Prefrontal Cortex (PFC): The front part of the cortex responsible for higher-order executive functions like decision-making, impulse control, and planning.
- Basal Ganglia: A group of deep brain structures (including the striatum and pallidum) that govern habit formation, routine behaviors, and the gating of actions via direct (“Go”) and indirect (“No-Go”) pathways.
- Cue-Induced Craving: Intense desire or urge to use a substance triggered by cues (places, objects, moods) previously associated with it. This craving is an inferred state – we measure it through self-report, behavior, or brain activity, since we cannot observe craving directly.
- Habit Formation: The process by which repeated behaviors become automatic, carried out with little conscious thought. In the brain, control over behavior shifts from deliberative cortical circuits to faster, stimulus-driven basal ganglia loops with repetition.
- “Go” vs “No-Go” Pathways: Two complementary circuits in the basal ganglia that regulate action initiation and inhibition. The direct (“Go”) pathway facilitates behavior, acting like a gas pedal that allows actions to proceed. The indirect (“No-Go”) pathway suppresses behavior, acting like a brake that helps prevent impulsive or inappropriate actions. Dopamine tends to bias this system toward action when reward cues are present.
Introduction: From Pavlov’s Dogs to Addictive Cues
In the early 1900s, the Russian physiologist Ivan Pavlov observed an intriguing phenomenon: dogs in his lab began to salivate not only when food was presented, but even before the food arrived – upon hearing the sound of a bell that had consistently been rung during feedings. This classic experiment demonstrated classical conditioning: a neutral stimulus (bell) became a powerful trigger for a physiological response (salivation) after repeated pairings with a reward (food). Figure 1 illustrates Pavlov’s setup, where over time the bell alone could elicit the dog’s hunger response after being associated with food.
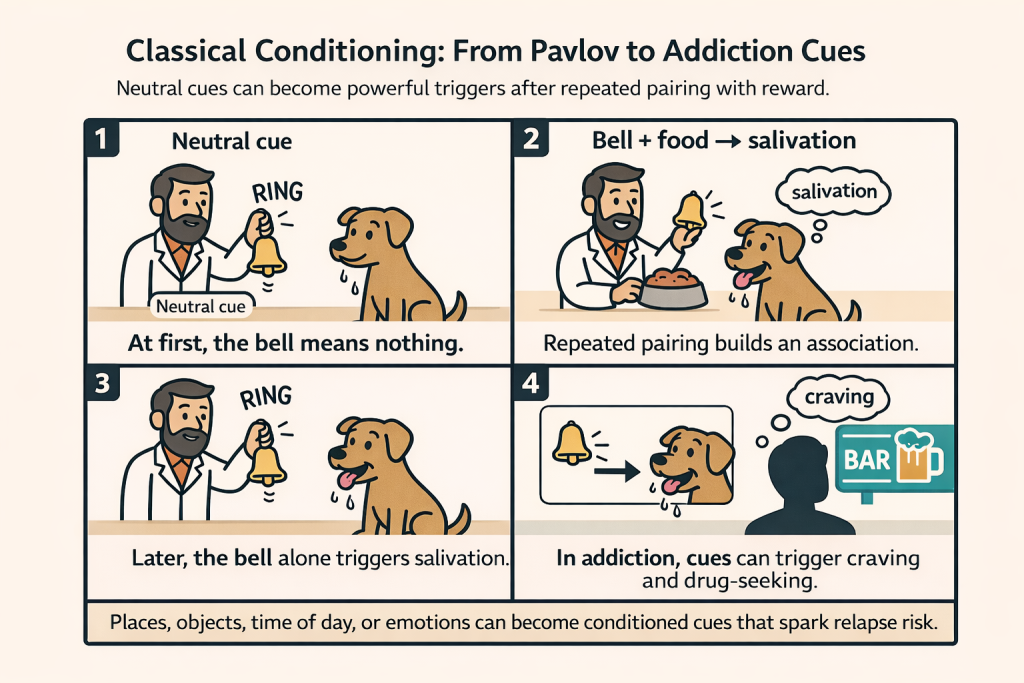
In addiction, we are all a bit like Pavlov’s dogs. Neutral things in the environment take on outsized meaning after being associated with drug use. A particular place (such as a bar or a friend’s house), an object (a pill bottle or a lighter), a certain time of day, or an emotional state (feeling stressed or lonely) can become strongly linked with the experience of using the substance. Through repeated pairings, these otherwise ordinary cues gain the power to set off intense craving. This learned connection between cue and craving is often called cue-induced craving, and it lies at the heart of why relapse is so common. People are constantly bombarded by reminders of their past drug use.
In a healthy brain, we normally have the self-control mechanisms to resist or redirect these urges. In addiction, those control systems weaken. Addiction thus reflects a multifaceted breakdown in “normal” brain function, a perfect storm of heightened motivation on one hand and impaired inhibition on the other. To understand this, we need to look “under the hood” at two interacting neural circuits: the limbic circuit, which assigns emotional significance and creates “wanting” for rewards or cues, and the cortico-basal ganglia circuit, which is responsible for decision-making, habits, and self-control. This circuit essentially determines whether or not a desire is acted upon.
In this module, we will first introduce the key brain regions in these circuits and their roles. We will then see how emotional cues and memories drive craving, and how, with repetition, drug-seeking behaviors become ingrained as habits. Finally, we will explore how chronic drug use alters these brain circuits, making cues even more potent while eroding our capacity to exert control, and how researchers study these changes. By the end, it will become clear how addiction emerges from the interaction of emotion, memory, and control. The sight or smell of a drug-related cue can spark a surge of desire. That surge is amplified by remembered pleasure and stress, and the normal “brakes” to stop the behavior fail to engage effectively.
Neuroanatomy of Addiction: Key Brain Regions and Circuits
A) Limbic “Reward” Circuit: Emotion and Memory Centers
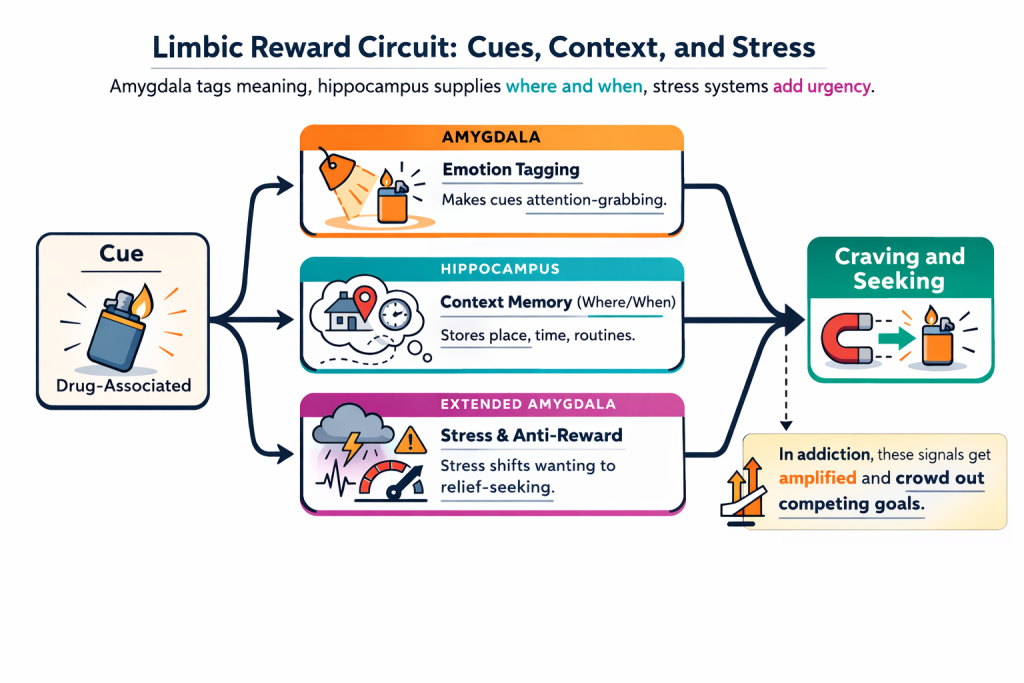
Amygdala: Emotional Tagging of Cues
The amygdala assigns emotional weight to events and their signals. Through Pavlovian conditioning, neutral cues paired with drug effects become attention-grabbing triggers. Later, the cue alone (e.g., a lighter, a bar sign) can spark anticipatory affect (e.g., excitement, desire, unease) that nudges behavior toward seeking.
Hippocampus: Context and “Where/When” Memory
The hippocampus records the setting: place, time, people, routines. These contextual details later reactivate memory and motivation. Returning to a “using context” (front porch at night, a certain commute) can rekindle craving because the context is now part of the learned association.
Extended Amygdala: Stress and the Anti-Reward System
The extended amygdala mediates negative affect, anxiety, irritability, dysphoria, especially during withdrawal or stress. With chronic use, stress neuromodulators (like CRF and dynorphin) ramp up, shifting motivation from seeking pleasure to seeking relief. Stress itself becomes a cue: “I feel bad → the drug will quiet this,” which powerfully amplifies craving and relapse risk.

How These Pieces Work Together
Amygdala tags what matters, hippocampus supplies where/when, and stress circuits add urgency under discomfort. These inputs converge on downstream motivation and action systems to prioritize cue-linked behaviors. In healthy learning, this keeps important signals from being ignored. In addiction, the same machinery overvalues drug-related cues and undercuts competing goals, laying the groundwork for compulsive pursuit.
B) Cortico-Basal Ganglia Circuit: Decision-Making and Self-Control Centers
If the limbic circuit generates motivational pull, the cortico-basal ganglia circuit determines whether that pull becomes action.
Prefrontal Cortex: Goal Setting and Brakes
The prefrontal cortex (PFC) supports planning, attention, and impulse control.
- Dorsolateral PFC (DLPFC): Holds rules and goals online (e.g., “skip dessert,” “stick to budget”) so they can guide choices.
- Orbitofrontal Cortex (OFC): Updates expected value, compares near-term payoff to longer-term costs and revises estimates with new outcomes.
- Anterior Cingulate Cortex (ACC): Detects conflict and errors (“this clashes with my goal”) and recruits additional control to course-correct.
Basal Ganglia & Striatum: Action Gatekeepers
Deep nuclei that decide whether an intended action is released or withheld.
- Direct (“Go”) pathway: Lifts the thalamic brake to facilitate a selected action.
- Indirect (“No-Go”) pathway: Reinforces the brake to suppress an action that shouldn’t proceed.
These pathways run in dynamic balance to keep behavior context-appropriate.
Dopamine: The Balance Shifter
Dopamine biases selection by enhancing “Go” via D1 and dampening “No-Go” via D2 receptors in striatal circuits. When cues predict reward, the scale tips toward action; when dopamine tone is low or outcomes look costly, restraint prevails.
Cortico–Basal Ganglia Loops: Parallel Control Channels
Cortex and basal ganglia communicate in loops that align intentions with actions.
- Motor loop: Automates well-practiced movements (motor cortex ↔ putamen).
- Associative/executive loop: Supports goal-directed choice (DLPFC ↔ caudate).
- Limbic loop: Integrates emotion and motivation into action selection (OFC/ACC ↔ ventral striatum).
These loops let goals shape habits, and let habits shape what feels easy to do.

Addiction: When Habits Overrun Control
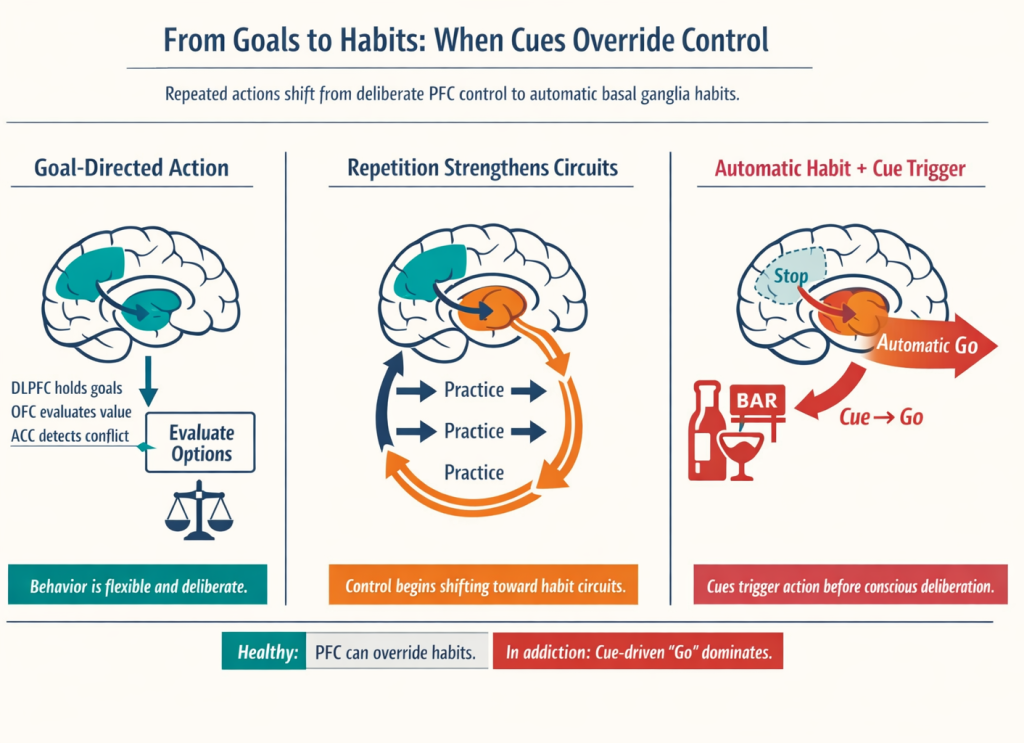
With repetition, drug seeking shifts from flexible, goal-directed decisions toward rigid, stimulus-bound habits in dorsal striatal circuits. Prefrontal “brakes” weaken, “Go” routines become default responses to cues and stress, and suppressing them requires disproportionate effort from control systems.
How These Pieces Work Together
PFC regions set goals and signal when more control is needed; basal ganglia decide whether an action is released or held back; dopamine tips the balance. In healthy behavior, this system balances desire with consequences. In addiction, chronic cue pairing and stress tilt the circuit toward automatic “Go,” undermining restraint and making drug-related actions hard to stop.

From Cue to Action: How Craving, Control, and Habits Interact
This section traces what happens in the brain from the moment a cue is detected to the moment a behavior is either enacted or inhibited. We examine first how the limbic circuit generates craving, then how the cortico-basal ganglia circuit selects or suppresses actions, and finally how these systems interact to determine whether desire becomes behavior.
A) Limbic circuit mechanism and flow: cue to craving
Cue-triggered craving starts as a learning story. Through repeated pairing with substance use, a previously neutral cue becomes a predictive signal, and that signal begins to carry motivational force.
To see the mechanism in motion, imagine a recovering alcoholic walking past a bar and suddenly feeling an overwhelming urge to drink, even though they have been sober for months. This is cue-induced craving, and it is driven by the limbic circuit.
- Cue detection (amygdala and hippocampus)
The cue is first recognized and interpreted. The amygdala tags the sensory input with emotional salience, for example a flash of excitement, nostalgia, or unease. In parallel, the hippocampus retrieves contextual details, such as “I have been here before” or “this is where I used to drink after work,” reactivating memories linked to the setting. - Motivational drive (nucleus accumbens and VTA dopamine)
Once the cue is recognized as meaningful, the limbic circuit engages the nucleus accumbens. Inputs from the amygdala and hippocampus provide the cue’s emotional weight and context. Midbrain dopamine neurons in the VTA increase firing and release dopamine into the nucleus accumbens, effectively preparing the brain for reward. This dopamine signal assigns incentive salience to the cue, so the cue becomes intensely “wanted” in the moment. - Physiological and affective response
Craving is not only cognitive. The body often responds with conditioned physiological changes, such as increased heart rate or salivation, similar to Pavlov’s dogs responding to the bell. Mood can shift as well, sometimes toward excitement, tension, or agitation, reflecting the limbic system gearing the organism toward drug seeking. - Stress amplification (extended amygdala, CRF and dynorphin)
If the person resists, or if they are already in withdrawal or stress, the extended amygdala can amplify the craving state. Stress neuromodulators like CRF and dynorphin contribute negative affect and a sense of urgency. The internal message becomes something like “All is not well until you get the drug,” which increases relapse risk by making non-use feel aversive.
Mini summary: what this circuit does in healthy learning, and what goes wrong in addiction
In healthy learning, this system helps us treat important cues as important. It tags predictive signals, retrieves relevant context, and mobilizes motivation so we can pursue adaptive rewards and avoid threats. In addiction, the same learning machinery becomes biased. Drug-associated cues become motivational magnets, craving becomes easier to trigger and harder to ignore, and stress states intensify the drive for relief. The limbic circuit does not simply remind someone of past use. It actively pulls attention, physiology, and motivation toward the drug.

B) Cortico–Basal Ganglia Mechanisms: Impulse to Action or Inhibition
The “Internal Committee” for Action Selection
The cortico‑basal ganglia circuit acts like your brain’s internal committee, deciding moment‑to‑moment which impulses should be turned into actions and which should be held back. It links frontal cortical regions (planning and value evaluation) with deep basal ganglia nuclei that gate motor and cognitive actions.
Go vs No‑Go Pathways: Balancing Actions
Within the basal ganglia, two opposing pathways control whether an intended action proceeds or is suppressed.
-
Direct (Go) pathway – the gas pedal: Striatal neurons expressing D1 receptors send inhibitory projections to the internal segment of the globus pallidus (GPi), which normally exerts a tonic brake on the thalamus. By inhibiting the GPi, the direct pathway disinhibits the thalamus and allows motor or cognitive signals from cortex to flow, facilitating the desired action.
-
Indirect (No‑Go) pathway – the brake pedal: Striatal neurons expressing D2 receptors project to the external globus pallidus (GPe) and the subthalamic nucleus (STN). These connections ultimately enhance GPi inhibition of the thalamus, reducing cortical activation and suppressing actions that should not occur. Because D2 receptors are inhibited by dopamine, this pathway becomes less active when dopamine levels are high.
A healthy brain maintains a dynamic balance between these pathways. The same system that quickly decides whether to stop or speed through a yellow light also governs social decisions and dietary choices.
Dopamine: The Great Modulator
Dopamine released from midbrain neurons (substantia nigra and ventral tegmental area) biases this balance. Dopamine excites D1‑expressing neurons in the direct pathway and inhibits D2‑expressing neurons in the indirect pathway. In other words, a dopamine surge functions like a motivational cheerleader saying “go for it,” making actions easier to initiate. When dopamine tone is low or outcomes look costly, the brake (indirect pathway) holds actions back.
Executive Control and Prefrontal Override
The prefrontal cortex (PFC) provides top‑down guidance to the basal ganglia. Different subregions support distinct control functions:
-
Dorsolateral prefrontal cortex (DLPFC): Often called the brain’s primary executive control center, it manages high‑level processes such as working memory, strategic planning, and cognitive flexibility. By holding goals in mind and evaluating consequences, the DLPFC can bias action selection toward long‑term objectives.
-
Orbitofrontal cortex (OFC): This region assigns value to potential rewards and punishments. Medial OFC neurons signal the subjective value of many rewards, whereas lateral OFC neurons represent non‑reward or punishment. Updating expected value is critical for deciding whether immediate pleasure outweighs long‑term costs.
-
Anterior cingulate cortex (ACC): The ACC monitors conflict and errors. It connects with limbic structures and is involved in emotion, autonomic responses, and selecting or initiating motor and cognitive actions. When a planned action clashes with a goal, the ACC recruits additional control to adjust behavior.
Through their projections to the striatum, these cortical regions can bias the Go/No‑Go competition. For example, if you decide to skip dessert, the DLPFC and ACC can engage the indirect pathway to suppress the impulse to grab cake, while promoting alternative actions like drinking water.
From Decisions to Habits: Shifting Loops
The basal ganglia contain multiple parallel loops: a “motor” loop linking motor cortex to putamen (automating well‑practiced movements), an “associative” loop linking DLPFC to caudate (supporting goal‑directed choices), and a “limbic” loop linking OFC/ACC to ventral striatum (integrating motivation and emotion). Early in learning, actions are goal‑directed and rely on the associative loop. With repetition, control gradually shifts to the motor loop, and behaviors become automatic habits. Research suggests that the dorsolateral striatum (putamen) is particularly involved in habitual actions, whereas the dorsomedial striatum (caudate) supports flexible, goal‑directed behavior. Moreover, experiments show that inactivating specific prefrontal areas can disrupt habitual responding and reinstate cognitive control.
Putting It Together
In everyday life, this cortico–basal ganglia system balances desires with consequences. Cortical regions set goals and evaluate outcomes, basal ganglia pathways gate actions, and dopamine tips the balance toward “Go” when cues predict reward. In addiction, repeated pairing of cues with drug rewards and stress can bias this system toward automatic “Go” responses, weaken prefrontal brakes, and entrench habits. Understanding these mechanisms helps explain why self‑control is harder during craving and why habits can persist despite conscious intentions to quit.
C) Integration: How the Two Circuits Interact
As someone with a history of substance use walks past a familiar cue (like a bar or pill bottle) two neural systems engage at once. The limbic circuit tags the cue with emotional significance and retrieves context, then engages the nucleus accumbens and dopaminergic neurons in the ventral tegmental area to generate a surge of motivational drive. At the same time, dopamine shifts the balance in the basal ganglia’s action-selection pathways, favoring the direct “Go” pathway over the indirect “No-Go” pathway.
Higher cortical regions try to assert control: the dorsolateral prefrontal cortex keeps goals in mind, the orbitofrontal cortex evaluates consequences, and the anterior cingulate cortex detects conflict. If these control signals are strong enough, they can bolster the “No-Go” pathway and redirect behavior. If they are not (such as in stressful situations) habitual responses stored in the dorsal striatum can take over.
The outcome depends on this balance. A cue-driven motivational pull, dopamine-biased gating, competing habits and deliberation, and the person’s current state together determine whether the individual turns into the bar or keeps walking.

How Chronic Substance Use Remaps the Brain’s Circuits
Chronic use of alcohol, opioids, stimulants, or cannabis can remap brain circuits by shifting the balance between cue- and stress-driven motivation (limbic systems and the ventral striatum) and executive control with action selection (prefrontal cortex and basal ganglia loops). Instead of a stable feedback loop between wanting and braking, repeated substance use sensitizes motivational circuits while impairing the brain’s ability to evaluate and inhibit actions.
From Reward Learning to Cue-Triggered Wanting
The amygdala tags experiences with emotional meaning, while the hippocampus encodes where and when those experiences occur. When a neutral cue, such as a location, object, or mood, is repeatedly paired with the rewarding effects of a drug, these regions store the association. Over time the nucleus accumbens (a key part of the ventral striatum) amplifies the “wanting” triggered by these cues through bursts of dopamine. This process, called incentive sensitization, means that a cue that used to be neutral now grabs attention and feels irresistible even if the pleasurable “liking” fades. Stress can further heighten this cue reactivity, making a simple reminder of past use provoke intense craving.
Stress, Withdrawal, and Negative Reinforcement
Alongside heightened “wanting,” chronic use activates the extended amygdala, a network that mediates stress and negative emotional states. During withdrawal or periods of abstinence this system generates anxiety, irritability, or dysphoria; using the substance temporarily relieves these feelings. This negative reinforcement loop encourages continued use not just to feel good but to avoid feeling bad. The repeated cycling between intoxication and withdrawal reshapes limbic circuits, making individuals more sensitive to stress and less responsive to natural rewards. While these changes can be profound, they reflect neuroplasticity and can partly reverse with sustained abstinence and supportive environments.
Why Self-Control Weakens and Habits Take Over
The prefrontal cortex (PFC) comprises several subregions that underlie working memory, valuation, and conflict monitoring. It communicates with the basal ganglia through “Go” and “No-Go” pathways. Repeated drug use alters this balance. As a result, impulses triggered by drug cues can more easily translate into action, while the cortical systems responsible for considering consequences and inhibiting behavior become less active. Over time, behavior shifts from being driven by goals to being controlled by automatic stimulus-response patterns. This is why drug-seeking can persist even when the individual no longer experiences significant pleasure.
Implications for Relapse Risk and Recovery
The interaction between enhanced cue- and stress-driven motivation and impaired executive control explains why relapse risk remains high. Stress or exposure to conditioned cues can trigger craving, while weakened self-control and entrenched habits make it hard to resist. Yet the same plasticity that allows these changes also offers hope. Treatments that reduce cue reactivity, regulate stress, and strengthen PFC functions, such as cognitive-behavioral therapy, mindfulness, medications, and environmental changes, can help restore balance. Building new, healthy routines can recruit the basal ganglia for alternative habits. Recognizing that addiction reflects learning and adaptation, not moral weakness, supports a compassionate approach and underscores that brain changes are partly reversible with treatment and supportive environments.
Common Circuit-Level Effects of Chronic Substance Use (General Patterns)
| Circuit component | Typical change with long-term use | Behavioral consequence |
|---|---|---|
| Amygdala & hippocampus | Enhanced emotional tagging and contextual memory of drug cues | Stronger cue-triggered craving and intrusive memories |
| Ventral striatum (nucleus accumbens) | Sensitized dopamine response; increased incentive salience | Heightened “wanting” for drug-related stimuli |
| Extended amygdala | Overactive stress and anti-reward signaling | Withdrawal anxiety and negative reinforcement of use |
| Prefrontal cortex (PFC) | Reduced activation and connectivity; hypofrontality | Impaired decision-making, valuation, and inhibition |
| Basal ganglia “Go” pathway | Stronger habit circuitry and action initiation | Faster initiation of drug-seeking actions |
| Basal ganglia “No-Go” pathway | Weaker inhibitory signaling | Difficulty suppressing impulses |
| White-matter tracts | Reduced integrity in frontal-striatal and limbic pathways | Slower communication and diminished top-down control |
Key takeaway: Long-term substance use sensitizes the brain’s reward and stress systems while weakening the circuits for self-control and habit regulation. This dual shift explains why cues and stress can trigger powerful cravings and why it can be so difficult to stop, but it also highlights the potential for recovery through interventions that restore balance and build new habits.
Studying an Addicted Brain: Imaging and Behavioral Tools
How do scientists and doctors actually see or measure the changes we’ve discussed in the living human brain? Advances in neuroimaging and cognitive testing provide several windows into the addicted brain:
- Functional MRI (fMRI) and cognitive tasks.
Researchers ask participants to perform tasks that engage self-control or respond to drug cues while in the scanner. The Stop-Signal Task (SST) requires people to press a button for a “Go” signal but stop when a “Stop” signal appears; healthy brains activate inferior frontal cortex and dorsal striatum during stopping, whereas people with addiction show reduced activation, indicating impulsivity. Another task, the n-back, engages working memory and attention; addicted individuals often show blunted dorsolateral prefrontal cortex activation. Cue-reactivity fMRI studies show that limbic regions such as the nucleus accumbens and amygdala light up when people with addiction see drug-related cues. - Structural MRI (sMRI).
High-resolution sMRI measures the size and shape of brain regions. Substance use disorders are associated with smaller hippocampal and amygdala volumes and thinning of the prefrontal cortex and orbitofrontal cortex. These structural changes correlate with functional deficits such as impulsivity. sMRI can also track recovery; some volumetric deficits partly normalize with abstinence. - Diffusion Tensor Imaging (DTI).
DTI maps the integrity of white-matter tracts by measuring fractional anisotropy. In addiction, frontal–striatal and limbic connections often show lower FA, indicating impaired communication highways, and reduced white-matter integrity is linked to higher relapse risk. Some recovery is observed with extended sobriety. - Positron Emission Tomography (PET).
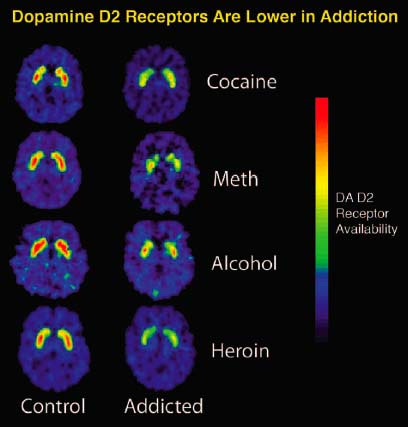
PET uses radioactive tracers to measure neurochemistry. It can assess dopamine D2 receptor availability (individuals with addictions have lower D2 receptor levels in the striatum than healthy controls) and measure dopamine release in response to cues. Since Module 8 already provides a detailed overview of PET methods and findings in addiction, this section notes only that PET supports downregulation of dopamine receptors and incentive sensitization. - Behavioral assessments.
Tasks such as go/no-go and decision-making tasks, along with questionnaires, quantify cognitive changes. Linking these behavioral measures to imaging findings connects brain changes to functional impairment.
Together, these tools provide complementary perspectives: fMRI shows functional activation patterns, sMRI and DTI reveal structural and connectivity changes, PET assesses neurochemical alterations, and behavioral tests link brain changes to real-world behaviors. They collectively demonstrate that addiction involves measurable brain alterations and that some aspects of brain function can recover with treatment and sustained abstinence.
Conclusion
The chapter synthesizes how chronic substance use fundamentally alters the balance between drive and control within the brain. Cues and stressors increasingly activate limbic structures such as the amygdala, hippocampus, and nucleus accumbens, making drug-related stimuli highly salient. At the same time, repeated exposure to substances erodes executive circuits in the prefrontal cortex and basal ganglia, weakening the “stop” systems that normally inhibit impulsive actions. These parallel changes mean that emotional and memory circuits push harder on behavior while self‑control circuits lose their regulatory grip. Recognizing addiction as a condition of heightened motivation coupled with impaired control underscores why treatment involves more than willpower: it requires managing cues and stress, strengthening healthy habits and alternative neural pathways, and sometimes using medications to stabilize brain chemistry. Encouragingly, neuroplasticity allows some recovery—dopamine receptor levels, frontal metabolism, and impulse control can improve with sustained abstinence and support.
These limbic and executive dynamics build on foundations laid in Module 8, which explored the mesolimbic dopamine pathway. That earlier module highlighted how dopamine drives “wanting” rather than “liking”; cravings can become exaggerated through incentive sensitization even as the pleasure of the drug fades. It also introduced the opponent‑process theory, showing how repeated dopamine surges tip the pleasure–pain balance toward dysphoria and craving. By integrating these reward concepts with the current module’s emphasis on stress, memory, and self‑control circuits, we see how excessive “wanting” (fueled by dopamine) is amplified by cue-induced limbic reactivity and unchecked by a weakened prefrontal system.

